The Chemistry of Water | Chemistry Basics
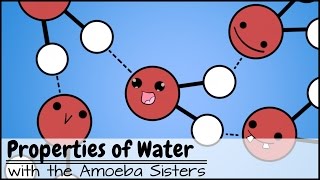
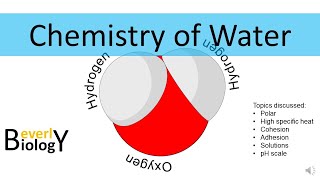
Water is a ubiquitous substance with some highly unusual chemical properties. Water molecules are polar in nature, which makes them interact with each other and other substances in fascinating ways. But why can salt dissolve in water when oil can’t? This episode of chemistry basics explores the chemical structure of water molecules, and gives you a chemical crash course in polarity and the molecular property that makes water so special – hydrogen bonds.
Subscribe! http://bit.ly/AmerChemSOc
Facebook! / chemmatters
Twitter! / acschemmatters
For more information, please visit our website: https://www.acs.org/content/acs/en/ed...
You might also like:
ChemMatters:
• ChemMatters: The Science Behind Calor...
Middle School Chemistry – Lesson Overviews:
• Molecules Matter
Kids Chemistry:
• Interactive Colors in Milk Chemist...
Produced by the American Chemical Society, the world’s largest scientific society. ACS is a global leader in providing access to chemistryrelated information and research through its multiple databases, peerreviewed journals and scientific conferences.
Join the American Chemical Society! https://bit.ly/Join_ACS